 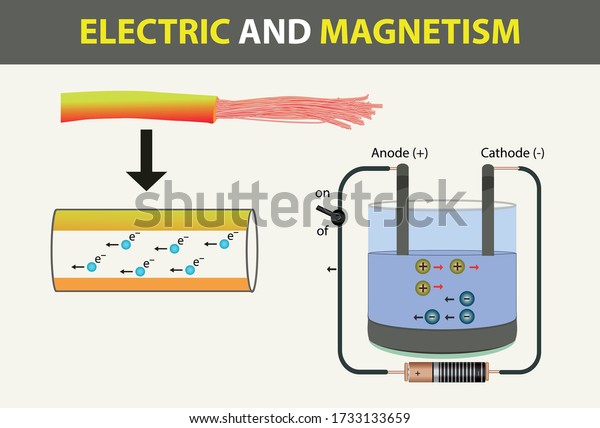
It is to be noted here that as electrons are moving from cathode to anode here thus the direction of the flow of current will be from anode towards the cathode. Here the positive electrode where oxidation is occurring is the anode and the electrode where reduction is occurring is the cathode. Similarly, the Cl – ions lose electrons ( oxidation) at the electrode connected with the negative terminal resulting in Cl 2 gas. On reaching the respective electrode, the battery potential allows the gaining of electrons ( reduction) by the Na+ ions, forming sodium metal. The electrode connected with the negative terminal of the battery attracts the Na + ions while the anions i.e., Cl – flows towards the electrode connected with the positive terminal. 
Along with this, the two electrodes are connected by a battery. In the molten state, Na+, and Cl – ions get separated and are in the free state. Here sodium chloride in the molten state is taken into which a pair of electrodes are immersed. Let us understand this by considering an electrolytic cell arrangement shown below: However, on considering the electrolytic cell, the anode and cathode terminal polarity will get reversed. However, the direction of the flow of current will be opposite to the flow of current.Īs you have noticed here that among the two electrodes, oxidation occurs at the Zinc electrode thus is anode with negative polarity and reduction occurs at the Copper electrode thus is cathode with positive polarity in a galvanic cell arrangement. The movement of anions (negatively charged particles) generates a current which flows through the metallic wire. Further, the switch is in the on state and we will get the closed-circuit arrangement then the electrons from the Zn electrode migrate ( oxidation) through the salt bridge and get reduced at the Cu electrode ( reduction). The two electrodes of zinc and copper that will act as anode and cathode are connected by a metallic wire through a switch.ĭuring the open condition of the switch, due to the open circuit arrangement, no reaction will occur in any of the beakers and so there will be no flow of current through the wire. To maintain electric contact between the two solutions a salt bridge containing potassium chloride is used. Here in the two separate beakers, we have copper sulphate and zinc sulphate solution. 
Hence, here the anode will attract positively charged particles and the cathode will attract the negatively charged particles.Ĭonsider a galvanic cell arrangement shown below in order to understand how the flow of current through the solution takes place. For a galvanic cell, anode holds the negative polarity whereas cathode acts as a positive terminal.Thus, an anode attracts negatively charged particles while a cathode attracts positively charged particles. For an electrolytic cell, anode acts as a positive terminal while a cathode holds negative polarity.This is so because the terminal polarity varies according to the type of cell i.e., electrolytic or galvanic in use. The specific denotation of the anode as positive and cathode as negative is wrong.While cathode corresponds to the electrode where reduction i.e., the gain of electrons occurs. The key factor of differentiation between anode and cathode is that anode corresponds to the electrode where oxidation i.e., loss of electrons occurs.Key Differences Between Anode and Cathode In electrolytic cell, it is a source of negative charge or electron donor. In electrolytic cell it is a source of positive charge or electron acceptor. Content: Anode Vs CathodeĪn anode in electrolytic cell attracts anions.Ī cathode in electrolytic cell attracts cations. when the electrode gains electrons which are released by the electrolyte undergoes reduction. However, when the reverse operation occurs i.e. It specifies the conductive phase where the transfer of charged carriers takes place.Īn electrode that loses electrons and is accepted by the electrolyte undergoes oxidation. More specifically, we can say, it is regarded as a surface where a redox reaction takes place between the metal and the solution.Įlectrode generally represents an electrical conductor/ semiconductor within the electrochemical cell. Electrode acts as a metallic contact through which the current enters and leaves the electrolyte. But first, see-Ī crucial component of an electrochemical cell that makes contact with electrolyte is known as an electrode. But in this content, you will get to know that the differentiation between anode and cathode is not merely done according to the type of polarity. People generally in misconception regards anode particularly as positive and cathode particularly as negative. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
